Breast biopsy or excision
Author:
Mikael Häggström [note 1]
Contents
Fixation
Generally 10% neutral buffered formalin. Breast specimens should be immersed for 6-72 hours.[1]
Comprehensiveness
On this resource, the following formatting is used for comprehensiveness:
- Minimal depth
- (Moderate depth)
- ((Comprehensive))
- Other legend
<< Decision needed between alternatives separated by / signs >>
{{Common findings / In case of findings}}
[[Comments]]
Link to another page
Gross processing
Selection and trimming
For excision (also called lumpectomy):
- Determine total specimen size. ((Weight the specimen[2]))
- Ink margins.Template:Ink note If sample orientations are marked, use different colors for different directions.[2]
- Palpate the specimen for masses. Compare with radiograph if available[2]
- Make 3-4 mm thick slices.[2]
- Submit:[2]
- Entire specimen if it can fit in 3-5 slices.
- If larger, 1 slice per cm of tumor (minimum of 3 slices of tumor), including both center and periphery of tumor.
- Additional suspicious areas, including those indicated by mammography
Breast specimens where breast cancer are possible should generally not be decalcified even when containing small calcifications, to preserve the ability to perform immunohistochemistry.
See also: General notes on gross processing
Gross report
- Size of original tissue sample, preferably in 3 dimensions.
- Description of inking
- Tumor properties, at least:
- (Description of sectioning and submissions.)
- ((Time of procurement and time of placement in formalin, in order to be able to calculate the cold ischemia time, which has effects on immunohistochemistry.[3]))
Example:
| (The specimen is received fresh and consists of) an irregular fragment of yellow tan fibrofatty tissue measuring 4.0 x 2.8 x 2.0 cm. (The specimen is oriented with two sutures and) the surgical margins are inked as follows: superior-blue, inferior-green, medial-red, lateral-yellow, anterior-orange, and posterior-black. {{A radioactive seed is embedded in the tissue.}} The tissue is serially sectioned {{to reveal a tan-gray, spiculated, indurated mass measuring _cm in greatest dimension. The mass is located _cm from the nearest (<<superior, inferior etc>>) margin. (The specimen is entirely submitted in sequential sections from medial to lateral in 10 cassettes. The medial and lateral margins are submitted as perpendicular sections.) The specimen was procured at _AM/PM on _/_/2020. The specimen was placed in formalin at _AM/PM on _/_/2020. |
Staining
Usually H&E staining.
Routine immunohistochemistry usually include estrogen and progesterone receptors (ER, PR) and HER2.[4]
Microscopic evaluation
If tumor is found, determine:
- Tumor size
- Malignancy
- Distance from excision margin
Malignancy
The most important is to classify a sample as either of the following:
- Benign
- Carcinoma in situ
- Invasive cancer
Most common types
| Finding | Relative incidence |
Histopathology | Image |
|---|---|---|---|
| Fibrocystic breast changes | 40% | ||
| No disease | 30% | ||
| Fibroadenoma | 7% | Proliferation of both stromal and epithelial components,[notes 1] arranged into either a pericanalicular pattern (stromal proliferation around epithelial structures), or an intracanalicular pattern (stromal proliferation compressing the epithelial structures into clefts). Fibroadenomas characteristically display hypovascular stroma compared to malignant tumors.[6][7][8] Furthermore, the epithelial proliferation appears in a single terminal ductal unit and has duct-like spaces surrounded by a fibroblastic stroma. The basement membrane is intact.[9] | 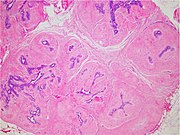
|
| Atypical ductal hyperplasia | 7%[10] | Epithelial proliferations which are not qualitatively or quantitatively abnormal enough to be classified as ductal carcinoma in situ.[10] | 
|
| Other benign mammary dysplasias and neoplasms | 5% | ||
| Breast cancer (in situ or invasive) | 10% | See next section. |
Breast cancer types
| Cancer type | Histopathology | Image |
|---|---|---|
| Invasive ductal carcinoma (IDC) | Carcinomatous cells are seen below the basement membrane of lactiferous ducts. Otherwise, there are no specific histologic characteristics, essentially making it a diagnosis of exclusion.[11] | 
|
| Ductal carcinoma in situ (DCIS) | Malignant epithelial cells confined to the ductal system of the breast, without invasion through the basement membrane.[12] | 
|
| Invasive lobular carcinoma (ILC) | The "classic" pattern is round or ovoid cells with little cytoplasm in a single-file infiltrating pattern, sometimes concentrically giving a targetoid pattern. | 
|
| Lobular carcinoma in situ (LCIS) |
Cells have indistinct cell borders, pale cytoplasm, and uniform small nuclei with evenly distributed chromatin and inconspicuous nucleoli.[13] |

|
| Mucinous carcinoma | Extracellular mucin areas around tumor cells. | 
|
| Medullary carcinoma | Seemingly fused tumor cells (syncytial pattern), and a prominent lymphoid infiltrate. | 
|
| Solid papillary carcinoma | Larger tumor nests with fibrovascular cores. | 
|
- Further information: Evaluation of tumors
If tumor is not found

Look for other abnormalities that may explain any diagnostic findings, mainly dense stromal fibrosis.
Microscopy report
A normal biopsy may be reported as follows
| (Fibrofatty tissue with benign ducts and lobules.) Negative for (atypia and) malignancy. |
Notes
- ↑ The proliferation of two histological components is called "biplasia", from Latin bis (“twice”) and -plasia (“formation”), or "biphasic proliferation"
- ↑ For a full list of contributors, see article history. Creators of images are attributed at the image description pages, seen by clicking on the images. See Patholines:Authorship for details.
Main page
References
- ↑ . Recommendations for HER2 Testing in Breast Cancer: ASCO – CAP Clinical Practice Guideline Update. College of American Pathologists (2013-10-17).
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 Monika Roychowdhury. Grossing (histologic sampling) of breast lesions. Pathologyoutlines.com. Topic Completed: 1 August 2012. Revised: 19 September 2019
- ↑ Khoury, Thaer (2018). "Delay to Formalin Fixation (Cold Ischemia Time) Effect on Breast Cancer Molecules ". American Journal of Clinical Pathology 149 (4): 275–292. doi:. ISSN 0002-9173.
- ↑ "Immunohistochemical detection of estrogen and progesterone receptor and HER2 expression in breast carcinomas: comparison of cell block and tissue block preparations ". Int J Clin Exp Pathol 2 (5): 476–80. 2009. PMID 19294006.
- ↑ Mitchell, Richard Sheppard; Kumar, Vinay; Abbas, Abul K.; Fausto, Nelson (2007). Robbins Basic Pathology (8th ed.). Philadelphia: Saunders. p. 739. ISBN 978-1-4160-2973-1.
- ↑ Tavassoli, F.A., ed (2003). World Health Organization Classification of Tumours: Pathology & Genetics: Tumours of the breast and female genital organs . Lyon: IARC Press. ISBN 978-92-832-2412-9.
- ↑ Jaya Ruth Asirvatham, M.B.B.S., Carlos C. Diez Freire, M.D., Cansu Karakas, M.D., Belinda Lategan, M.D., Nat Pernick, M.D., Emily S. Reisenbichler, M.D., Monika Roychowdhury, M.D., Mary Ann Gimenez Sanders, M.D, Ph.D., Gary Tozbikian, M.D., Hind Warzecha, M.D. Senior Authors: Julie M. Jorns, M.D., Shahla Masood. Breast nonmalignant, Fibroepithelial neoplasms, Fibroadenoma. Retrieved on 2019-11-04. Revised: 31 October 2019
- ↑ Rosen, PP. (2009). Rosen's Breast Pathology (3rd ed.). ISBN 978-0-7817-7137-5.
- ↑ . Fibroadenoma of the breast.
- ↑ 10.0 10.1 David J. Myers; Andrew L. Walls.. Atypical Breast Hyperplasia. StatPearls, National Center for Biotechnology Information. Last Update: February 15, 2019.
- ↑ Peter Abdelmessieh. Breast Cancer Histology. Medscape. Retrieved on 2019-10-04. Updated: May 24, 2018
- ↑ Siziopikou, Kalliopi P. (2013). "Ductal Carcinoma In Situ of the Breast: Current Concepts and Future Directions ". Archives of Pathology & Laboratory Medicine 137 (4): 462–466. doi:. ISSN 0003-9985.
- ↑ 13.0 13.1 Sucheta Srivastava. Breast - Noninvasive lobular neoplasia - LCIS classic. Topic Completed: 1 September 2017. Minor changes: 21 June 2020
- ↑ Nassar, Lara; Baassiri, Amro; Salah, Fatima; Barakat, Andrew; Najem, Elie; Boulos, Fouad; Berjawi, Ghina (2019). "Stromal Fibrosis of the Breast: A Spectrum of Benign to Malignant Imaging Appearances ". Radiology Research and Practice 2019: 1–6. doi:. ISSN 2090-1941.
Image sources
