Difference between revisions of "Evaluation of suspected malignancies"
(→Overall diagnosis: Better) |
m (br) |
||
| (20 intermediate revisions by the same user not shown) | |||
| Line 1: | Line 1: | ||
| − | {{Top | + | <noinclude>{{Top |
|author1=[[User:Mikael Häggström|Mikael Häggström]] | |author1=[[User:Mikael Häggström|Mikael Häggström]] | ||
|author2= | |author2= | ||
| − | }} | + | }}</noinclude> |
| − | For [[evaluation]] of '''suspected malignancies''' such as '''tumors''', the most important aspect is whether it is benign or malignant. If malignant, then staging is necessary.<ref name=cancer>{{cite web |url= http://www.cancer.gov/cancertopics/factsheet/detection/staging |title=Cancer staging |date= |publisher=National Cancer Institute |accessdate=4 January 2013}}</ref> There are generally specific criteria for various forms of tumors, which should be used whenever applicable, but following are some generalizations. | + | For [[evaluation]] of '''suspected malignancies''' such as '''[[tumors]]''', the most important aspect is whether it is benign or malignant. If malignant, then staging is necessary.<ref name=cancer>{{cite web |url= http://www.cancer.gov/cancertopics/factsheet/detection/staging |title=Cancer staging |date= |publisher=National Cancer Institute |accessdate=4 January 2013}}</ref> There are generally specific criteria for various forms of tumors, which should be used whenever applicable, but following are some generalizations. |
| − | A general approach is to start looking at | + | A general approach is to start looking at a slide which seems to contain non-necrotic tumor, and if possible it should also show surrounding non-tumor tissue, so that the interface can be appreciated (and tumors are generally less necrotic at the periphery). |
==Benign or malignant== | ==Benign or malignant== | ||
| − | |||
{|class="wikitable" | {|class="wikitable" | ||
! !! Benign<ref name=amboss>{{cite web|url=https://www.amboss.com/us/knowledge/General_oncology|title=General oncology|website=Amboss|accessdate=2020-01-29}}</ref> !! Malignant<ref name=amboss/> | ! !! Benign<ref name=amboss>{{cite web|url=https://www.amboss.com/us/knowledge/General_oncology|title=General oncology|website=Amboss|accessdate=2020-01-29}}</ref> !! Malignant<ref name=amboss/> | ||
| Line 30: | Line 29: | ||
*Numerous mitotic figures | *Numerous mitotic figures | ||
|} | |} | ||
| + | <gallery mode=packed heights=220> | ||
| + | File:Normal and cancer cells structure.jpg|Cellular features of malignant cells versus normal cells. | ||
| + | File:Nuclear-to-cytoplasm ratios.png|An increased nucleus-cytoplasm ratio is generally an indication of malignancy. | ||
| + | </gallery> | ||
| + | {{Primary tumor versus metastasis}} | ||
| + | <br>{{further|Metastasis}} | ||
| + | ==Histopathologic type== | ||
| + | For specific diagnoses by organ system, see anatomic diagram on Patholines '''[[Main page]]'''. This resource will give the main steps towards reaching a diagnosis, but before making a tumor diagnosis, generally be sure that it fulfills the criteria of the condition according to [https://publications.iarc.fr/Book-And-Report-Series/Who-Classification-Of-Tumours The WHO Classification of tumors], and generally consult an experienced pathologist as well until you feel confident. | ||
| − | + | Visually, tumors and other suspected malignancies can usually be classified into one of the following groups: | |
| − | |||
| − | |||
| − | Visually, tumors and other suspected malignancies can | ||
<gallery mode=packed heights=220> | <gallery mode=packed heights=220> | ||
File:Histopathology of endometrioid cancer, grade 1, nuclear grade 2.jpg|'''Gland-like tumors''', with tumor cells around clearings | File:Histopathology of endometrioid cancer, grade 1, nuclear grade 2.jpg|'''Gland-like tumors''', with tumor cells around clearings | ||
| + | File:Histopathology of squamous-cell carcinoma of the lung.jpg|'''Squamoid tumors''', typically having abundant eosinophilic cytoplasm.<ref name="pmid32316685">{{cite journal| author=Choi JH, Ro JY| title=Epithelioid Cutaneous Mesenchymal Neoplasms: A Practical Diagnostic Approach. | journal=Diagnostics (Basel) | year= 2020 | volume= 10 | issue= 4 | pages= | pmid=32316685 | doi=10.3390/diagnostics10040233 | pmc=7236000 | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=32316685 }} </ref> | ||
File:Histopathology of an ovarian fibroma.jpg|'''Spindle-cell tumors''', with elongated cells and/or nuclei | File:Histopathology of an ovarian fibroma.jpg|'''Spindle-cell tumors''', with elongated cells and/or nuclei | ||
| − | |||
File:Histopathology of Merkel-cell carcinoma with undifferentiated tumor cells.png|'''Undifferentiated tumors''', with few to no visual clues to their origin. | File:Histopathology of Merkel-cell carcinoma with undifferentiated tumor cells.png|'''Undifferentiated tumors''', with few to no visual clues to their origin. | ||
</gallery> | </gallery> | ||
| Line 44: | Line 48: | ||
Further pinpointing of a specific tumor type is often attained by thinking of one or more possible diagnoses, and looking up their '''differential diagnoses''', followed by comparing their microscopic descriptions and multiple micrographs with the case at hand. When two or more diagnoses seem to fit with the case at hand, consider performing '''immunohistochemistry'''. Find relevant target proteins that are expected to stain substantially differently between the possible diagnoses. If it's not evident from initial sources, you may use Immunoquery.com which will generally suggest the most relevant target proteins to distinguish the suspected conditions at hand. | Further pinpointing of a specific tumor type is often attained by thinking of one or more possible diagnoses, and looking up their '''differential diagnoses''', followed by comparing their microscopic descriptions and multiple micrographs with the case at hand. When two or more diagnoses seem to fit with the case at hand, consider performing '''immunohistochemistry'''. Find relevant target proteins that are expected to stain substantially differently between the possible diagnoses. If it's not evident from initial sources, you may use Immunoquery.com which will generally suggest the most relevant target proteins to distinguish the suspected conditions at hand. | ||
| − | + | ===Gland-like tumors=== | |
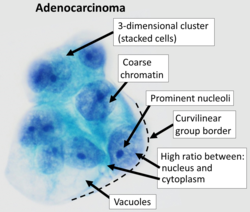
| + | [[File:Pap stain of adenocarcinoma in peritoneal fluid.png|thumb|250px|Typical features of adenocarcinomas on cytology (Pap stain). Vacuoles may be seen in both mucinous and serous tumors.]] | ||
Gland-like tumors are mainly evaluated for cellular atypia, architectural dysplasia and invasion, and thereby classified into the following main categories: | Gland-like tumors are mainly evaluated for cellular atypia, architectural dysplasia and invasion, and thereby classified into the following main categories: | ||
*'''Hyperplastic''' lesions, lacking significant atypia | *'''Hyperplastic''' lesions, lacking significant atypia | ||
| Line 56: | Line 61: | ||
:*[[Endometrial adenocarcinoma]] | :*[[Endometrial adenocarcinoma]] | ||
| − | ====Spindle-cell tumors | + | ===Squamoid tumors=== |
| + | These are more or less looking like a '''[[squamous-cell carcinoma]]''': | ||
| + | <gallery mode=packed heights=220> | ||
| + | File:Histopathology of squamous-cell carcinoma.png|Main characteristics of squamous-cell carcinoma | ||
| + | File:Histopathology of invasive squamous cell carcinoma.jpg|Typical well-differentiated nests have cells with abundant eosinophilic cytoplasm. Keratinizing centers are seen as well. | ||
| + | </gallery> | ||
| + | Differential diagnoses depend on location, such as: | ||
| + | *'''[[Squamous-cell carcinoma of the skin]]''', with '''[[Template:Squamous-cell like skin proliferations - differential diagnosis|multiple differential diagnoses]]''' | ||
| + | *'''[[Squamous-cell carcinoma of the lung]]''' | ||
| + | *Urinary bladder: '''[[Urothelial versus squamous-cell carcinoma]]''' | ||
| + | *Cervix uteri: '''[[Cervical dysplasia]]''' | ||
| + | |||
| + | ===Spindle-cell tumors=== | ||
For '''Spindle-cell tumors''', the shape of the nuclei is a clue to the diagnosis, with the following tendency: | For '''Spindle-cell tumors''', the shape of the nuclei is a clue to the diagnosis, with the following tendency: | ||
*Pointed on both ends: True fibroblastic tumors | *Pointed on both ends: True fibroblastic tumors | ||
| Line 62: | Line 79: | ||
*Blunted on both ends ("cigar-shaped"): Smooth muscle | *Blunted on both ends ("cigar-shaped"): Smooth muscle | ||
*Triangular: Myofibroblastic | *Triangular: Myofibroblastic | ||
| − | Evaluate specifically by location when possible | + | Evaluate specifically by location when possible<noinclude>: |
| + | *'''[[Spindle-cell tumors of the midgut]]''' | ||
| + | *'''[[Soft tissue tumor]]'''</noinclude> | ||
| + | |||
| + | ===Further histopathologic subtyping and grading=== | ||
| + | Beyond determining overall malignancy diagnosis (such as adenocarcinoma), probable origin and staging, classification of tumors into a specific histopathologic type or grade is generally of relatively less value. In cases of clearly non-malignant tumors where it is difficult to determine the specific histopathologic type or grade, it is generally acceptable to conclude the evaluation and report it as such, unless the clinician specifically requests otherwise. For potentially malignant or high-risk tumors, typing and grading often still affects the management. | ||
| − | + | ===Undifferentiated tumor=== | |
| − | An initial panel of cytokeratin (CK), S100, vimentin and LCA (CD45) can be used.<ref name="pmid25427040">{{cite journal| author=Lin F, Liu H| title=Immunohistochemistry in undifferentiated neoplasm/tumor of uncertain origin. | journal=Arch Pathol Lab Med | year= 2014 | volume= 138 | issue= 12 | pages= 1583-610 | pmid=25427040 | doi=10.5858/arpa.2014-0061-RA | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=25427040 }} </ref> | + | An initial panel of cytokeratin (CK), S100, vimentin and LCA (CD45) can be used (see source article for subsequent work-up).<ref name="pmid25427040">{{cite journal| author=Lin F, Liu H| title=Immunohistochemistry in undifferentiated neoplasm/tumor of uncertain origin. | journal=Arch Pathol Lab Med | year= 2014 | volume= 138 | issue= 12 | pages= 1583-610 | pmid=25427040 | doi=10.5858/arpa.2014-0061-RA | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=25427040 }} </ref> |
| − | === | + | ===Non-neoplastic=== |
| − | If a '''neoplasm has been ruled out''' for what clinically appeared like a tumor, seek a diagnosis that can be consistent with | + | If a '''neoplasm has been ruled out''' for what clinically appeared like a tumor, seek a diagnosis that can be consistent with the clinical findings that caused the suspicion. If no explanation is found on the slides, generally take additional levels on the paraffin block, or more sections from any leftover tissue. |
| + | |||
| + | For example, for a breast biopsy of what appeared to look like a mass, and there is no neoplasia, look mainly for dense fibrosis or other fibrous changes, so that you can report it and thereby explain the finding, rather than merely writing "benign breast tissue". | ||
| + | |||
| + | ===Heterogeneity=== | ||
| + | After having characterized a suspected malignancy, still screen through it for any significant areas that are different and may need own mentioning, or even change the overall type or grade. | ||
==Additional levels or slices== | ==Additional levels or slices== | ||
| − | + | Situations requiring additional material include mainly where tumor is expected but nevertheless not seen on existing slides. Such cases include: | |
| + | *The gross report or other observation describes a tumor or polyp, but none is seen on microscopy. | ||
*Re-excision does not identify tumor cells in a clearly non-radical primary excision or biopsy. | *Re-excision does not identify tumor cells in a clearly non-radical primary excision or biopsy. | ||
| − | + | ||
| + | Also consider more material if the most aggressive pattern is seen in the last available section, in which case more sections are indicated (from the same paraffin block if additional tissue is not available). | ||
| + | |||
Depending on availability and greatest suspicion, additional material is either acquired by taking addition step sections of remaining tissue in a paraffin block, or taking additional slices from the original specimen. | Depending on availability and greatest suspicion, additional material is either acquired by taking addition step sections of remaining tissue in a paraffin block, or taking additional slices from the original specimen. | ||
| Line 91: | Line 121: | ||
** N2: tumor spread to an extent between N1 and N3 (N2 is not used at all sites) | ** N2: tumor spread to an extent between N1 and N3 (N2 is not used at all sites) | ||
** N3: tumor spread to more distant or numerous regional lymph nodes (N3 is not used at all sites) | ** N3: tumor spread to more distant or numerous regional lymph nodes (N3 is not used at all sites) | ||
| − | '''M''': presence of distant metastasis | + | '''M''': presence of distant metastasis {{Further|Metastasis}} |
** M0: no distant metastasis | ** M0: no distant metastasis | ||
** M1: metastasis to distant organs (beyond regional lymph nodes) | ** M1: metastasis to distant organs (beyond regional lymph nodes) | ||
| Line 108: | Line 138: | ||
File:Histopathology of typical micropapillary urothelial carcinoma infiltrating the lamina propria - crop.jpg|If unsure whether a case is true lymphovascular invasion or a retraction artifact (imaged), use immunohistochemistry for D2-40 to highlight lymphatic vessels or CD-31 for blood vessels. | File:Histopathology of typical micropapillary urothelial carcinoma infiltrating the lamina propria - crop.jpg|If unsure whether a case is true lymphovascular invasion or a retraction artifact (imaged), use immunohistochemistry for D2-40 to highlight lymphatic vessels or CD-31 for blood vessels. | ||
</gallery> | </gallery> | ||
| − | |||
| − | |||
[[File:Histopathology of pancreatic adenocarcinoma with treatment effect.jpg|thumb|Also note "treatment effect", seen as fibroelastotic tissue, here with scattered remaining tumor cells.]] | [[File:Histopathology of pancreatic adenocarcinoma with treatment effect.jpg|thumb|Also note "treatment effect", seen as fibroelastotic tissue, here with scattered remaining tumor cells.]] | ||
| − | |||
==Reporting== | ==Reporting== | ||
{{CAP}} | {{CAP}} | ||
| + | If a surgery produces a specimen with cancer, as well as re-excisions from certain directions, you should preferably give the closest distance to margins in each specimen, as well as the closest distance overall in a synoptic, for example: | ||
| + | {|class=wikitable | ||
| + | | '''A. (Specimen with most of the cancer)''' | ||
| + | *(...) | ||
| + | *Invasive carcinoma is present at inked medial margin (see specimen B for final medial margin), and is located 0.3 cm from the lateral margin. | ||
| + | |||
| + | '''B. (Re-excision in the direction of the medial margin)''' | ||
| + | *(...) | ||
| + | *Invasive carcinoma is located 0.5 cm from the new medial margin. | ||
| + | |||
| + | '''Synoptic report''' | ||
| + | *(...) | ||
| + | *Distance from invasive carcinoma to closest margin: 0.3 cm | ||
| + | *Closest margin(s) to invasive carcinoma: Lateral | ||
| + | |} | ||
{{Reporting}} | {{Reporting}} | ||
<noinclude> | <noinclude> | ||
Revision as of 11:28, 17 November 2022
Author:
Mikael Häggström [note 1]
For evaluation of suspected malignancies such as tumors, the most important aspect is whether it is benign or malignant. If malignant, then staging is necessary.[1] There are generally specific criteria for various forms of tumors, which should be used whenever applicable, but following are some generalizations.
A general approach is to start looking at a slide which seems to contain non-necrotic tumor, and if possible it should also show surrounding non-tumor tissue, so that the interface can be appreciated (and tumors are generally less necrotic at the periphery).
Contents
Benign or malignant
| Benign[2] | Malignant[2] | |
|---|---|---|
| Gross examination |
|
Possibly:
|
| Microscopy | Almost no irregularities of cellular structures | Nuclear atypia:
|
Primary tumor versus metastasis

Indications of a metastasis rather than primary tumor are mainly:
- Tumors that are unlikely to arise at the location at hand.
- Tumors conforming to more likely metastasis pathways.
If a suspected malignancy is present, generally check the patient history for any history of cancer, especially for tumors in more common metastasis sites, which mainly include lung, bone, liver and/or brain. In case of such history, preferably look at the microscopy slides of the past cancer to help determining whether the current case is of the same origin, versus a primary at the current body location, versus a metastasis of yet another location. If there is no known history of cancer, still consider a metastasis of unknown primary origin, especially for suspected malignancies in lymph nodes, liver, lungs, bones, or skin.[4]
Further information: Metastasis
Histopathologic type
For specific diagnoses by organ system, see anatomic diagram on Patholines Main page. This resource will give the main steps towards reaching a diagnosis, but before making a tumor diagnosis, generally be sure that it fulfills the criteria of the condition according to The WHO Classification of tumors, and generally consult an experienced pathologist as well until you feel confident.
Visually, tumors and other suspected malignancies can usually be classified into one of the following groups:
Squamoid tumors, typically having abundant eosinophilic cytoplasm.[5]
Further pinpointing of a specific tumor type is often attained by thinking of one or more possible diagnoses, and looking up their differential diagnoses, followed by comparing their microscopic descriptions and multiple micrographs with the case at hand. When two or more diagnoses seem to fit with the case at hand, consider performing immunohistochemistry. Find relevant target proteins that are expected to stain substantially differently between the possible diagnoses. If it's not evident from initial sources, you may use Immunoquery.com which will generally suggest the most relevant target proteins to distinguish the suspected conditions at hand.
Gland-like tumors
Gland-like tumors are mainly evaluated for cellular atypia, architectural dysplasia and invasion, and thereby classified into the following main categories:
- Hyperplastic lesions, lacking significant atypia
- Adenomas, which can range from mild to high-grade dysplastic, yet are generally confined within their anatomic layers, that is, they are not invasive.
- Adenocarcinomas, with the main criterion being invasiveness. Evaluate specifically by location when possible. Some specific locations included in this resource:
Squamoid tumors
These are more or less looking like a squamous-cell carcinoma:
Differential diagnoses depend on location, such as:
- Squamous-cell carcinoma of the skin, with multiple differential diagnoses
- Squamous-cell carcinoma of the lung
- Urinary bladder: Urothelial versus squamous-cell carcinoma
- Cervix uteri: Cervical dysplasia
Spindle-cell tumors
For Spindle-cell tumors, the shape of the nuclei is a clue to the diagnosis, with the following tendency:
- Pointed on both ends: True fibroblastic tumors
- Pointed on one end and blunted on the other ("bullet-shaped"): Neural
- Blunted on both ends ("cigar-shaped"): Smooth muscle
- Triangular: Myofibroblastic
Evaluate specifically by location when possible:
Further histopathologic subtyping and grading
Beyond determining overall malignancy diagnosis (such as adenocarcinoma), probable origin and staging, classification of tumors into a specific histopathologic type or grade is generally of relatively less value. In cases of clearly non-malignant tumors where it is difficult to determine the specific histopathologic type or grade, it is generally acceptable to conclude the evaluation and report it as such, unless the clinician specifically requests otherwise. For potentially malignant or high-risk tumors, typing and grading often still affects the management.
Undifferentiated tumor
An initial panel of cytokeratin (CK), S100, vimentin and LCA (CD45) can be used (see source article for subsequent work-up).[6]
Non-neoplastic
If a neoplasm has been ruled out for what clinically appeared like a tumor, seek a diagnosis that can be consistent with the clinical findings that caused the suspicion. If no explanation is found on the slides, generally take additional levels on the paraffin block, or more sections from any leftover tissue.
For example, for a breast biopsy of what appeared to look like a mass, and there is no neoplasia, look mainly for dense fibrosis or other fibrous changes, so that you can report it and thereby explain the finding, rather than merely writing "benign breast tissue".
Heterogeneity
After having characterized a suspected malignancy, still screen through it for any significant areas that are different and may need own mentioning, or even change the overall type or grade.
Additional levels or slices
Situations requiring additional material include mainly where tumor is expected but nevertheless not seen on existing slides. Such cases include:
- The gross report or other observation describes a tumor or polyp, but none is seen on microscopy.
- Re-excision does not identify tumor cells in a clearly non-radical primary excision or biopsy.
Also consider more material if the most aggressive pattern is seen in the last available section, in which case more sections are indicated (from the same paraffin block if additional tissue is not available).
Depending on availability and greatest suspicion, additional material is either acquired by taking addition step sections of remaining tissue in a paraffin block, or taking additional slices from the original specimen.
Staging
Staging is generally done by TNM classification. Specific TNM systems should be used whenever applicable, mainly the manual by the American Joint Committee on Cancer (AJCC) if you can access it. Further information: Secrets . Otherwise, a general system may be used:[1]
|
T: size or direct extent of the primary tumor
N: degree of spread to regional lymph nodes
M: presence of distant metastasis Further information: Metastasis
|
Put your main focus on features that will determine the final stage. For example, if you see a lymph node involved by cancer, the presence or absence of lymphatic invasion is no longer critical, but rather the presence or absence of additional involved nodes or distant metastasis.
Radicality

Determine if malignant cells are located close to, or even in, any surgical resection margins.
Lymphovascular invasion
Lymphovascular invasion should always be mentioned. When present at margins, it does not count as tumor extension.
Reporting
For cancers, generally include a synoptic report, such as per College of American Pathologists (CAP) protocols at cap.org/protocols-and-guidelines. If a surgery produces a specimen with cancer, as well as re-excisions from certain directions, you should preferably give the closest distance to margins in each specimen, as well as the closest distance overall in a synoptic, for example:
A. (Specimen with most of the cancer)
B. (Re-excision in the direction of the medial margin)
Synoptic report
|
See also: General notes on reporting
|
Further reading: |
Notes
- ↑ For a full list of contributors, see article history. Creators of images are attributed at the image description pages, seen by clicking on the images. See Patholines:Authorship for details.
Main page
References
- ↑ 1.0 1.1 . Cancer staging. National Cancer Institute. Retrieved on 4 January 2013.
- ↑ 2.0 2.1 . General oncology. Amboss. Retrieved on 2020-01-29.
- ↑ List of included entries and references is found on main image page in Commons: Wikimedia Commons: Metastasis sites for common cancers.svg
- ↑ Lymph nodes, liver, lungs, bones, or skin are the main sites of cancer of unknown primary origin (CUP):
. Cancer of Unknown Primary Origin. Memorial Sloan Kettering Cancer Center. Retrieved on 20222-10-14. - ↑ Choi JH, Ro JY (2020). "Epithelioid Cutaneous Mesenchymal Neoplasms: A Practical Diagnostic Approach. ". Diagnostics (Basel) 10 (4). doi:. PMID 32316685. PMC: 7236000. Archived from the original. .
- ↑ Lin F, Liu H (2014). "Immunohistochemistry in undifferentiated neoplasm/tumor of uncertain origin. ". Arch Pathol Lab Med 138 (12): 1583-610. doi:. PMID 25427040. Archived from the original. .
Image sources