Starting pathology (entire handbook)

Author: Mikael Häggström, M.D.
This is a handbook directed at new pathology trainees (mainly residents or trainees with medical doctor degrees or similar).
Contents
- 1 Copies and prints
- 2 Contact
- 3 Learning pathology
- 4 What a pathologist needs to memorize
- 5 Using resources
- 6 Test question
- 7 Using this resource
- 8 Using question banks
- 9 Learning clinical pathology
- 10 General advice
- 11 Emergent pathology
- 12 Fixation
- 13 Gross processing
- 14 Evaluation of tumors
- 15 Reporting
- 16 Notes
- 17 References
Copies and prints
To get a printable version of this resource, go to patholines.org, click Starting pathology (handbook), and click Printable version in the left hand menu. For best print results, use a color printer and color printer paper, or google a "copy shop" for a color print delivery or in-store pickup (possibly as a photo book, otherwise you can keep them in a binder).
This resource is released under the Creative Commons Attribution 4.0 International (CC-BY 4.0) license, meaning that if you reuse any of its content (such as in scholarly articles), you need to mention its author (Mikael Häggström, M.D.). Also, many images are Public Domain, that is, without any restrictions. The status of each image is seen on its description page (found by clicking the image at Patholines.org). Furthermore, tables and graphs are never copyrightable unless they have some artistic, literary or other copyrightable aspect.[1]Further information: Patholines:Copyright
Contact
To contact the page creator/author, write a message to mikael![]() haeggstroem
haeggstroem![]() nuvancehealth
nuvancehealth![]() org.
org.
Please give feedback on any errors that you encounter in this resource, as well as pertinent omissions that you feel should be there. Preferably, contribute yourself: See the Contribute page at Patholines.org.
Learning pathology
The goal of this resource is to make a new pathology trainee able to properly handle at least 70% of cases that are expected at an average general pathology department, including the exclusion of the most pertinent differential diagnoses thereof. Its aim is to present you at least one way of doing things that is at least adequate for a particular situation, so that although there may be various routines at various locations, adherence to these methods should not result in a worse reprimand from a senior than "although that was adequate, that is not how we do it here". Still ask for help whenever needed, such as first time you are doing something, or whenever you are not sure about what to do, especially when doing something potentially irreversible.
What a pathologist needs to memorize
The best method for memorization is generally through repeated exposure in everyday practice, and the scope thereof will depend on your eventual location and subspecialty, and you will eventually forget everything else, more or less. Yet, the following things are most important for a pathologist trainee to focus on memorizing:
- Emergency pathology, mostly relating to intraoperative or frozen section consultations. This includes information that usually cannot be timely looked up on the Internet when needed.
- Main pitfalls: The most common and dangerous situations where a pathologist may not recognize the need to look something up further or ask a senior colleague, or where a significant risk of diagnosis error remains after doing so.
- Interpretation of non-written results, including patterns and signs which can be seen grossly or under the microscope. It confers the ability to translate visuals and other more or less non-verbalizable information into words that can be looked up if needed, and conceive the most likely diagnoses based on both describable and more abstract appearances.
- Proficiency in diagnosing equivocal or borderline cases where readily available sources and evidence usually deal with discrete and specific disease entities and subcategories thereof. Unusual or equivocal presentations of very common diseases and conditions are still generally more common than rare diseases, and constitutes a major workload in everyday pathology practice. However, most textbooks still give disproportionately large room for rare diseases compared to such presentations. Nevertheless, strive to master the common conditions (including the most common pitfalls) first, and skip the uncommon in the meantime. After all, you will get by with effectively handling the regular cases wherever you work, so that when unfamiliar cases appear, you will have time to study or ask others for how to handle them, as long as you know your pitfalls, and have an idea about your unknowns:
- Having an idea of one’s unknowns; being aware of a lack of knowledge in unfamiliar fields. For example, a pathologist generally does not need in depth knowledge about diseases that are generally sent out to specialized centers (such as pediatric musculoskeletal oncology), but just enough to be able to suspect such diseases. In the same manner, a pathologist needs to have an idea of the optimal limits of practice, between what should optimally be handled personally by the pathologist and what is better handled by others, including senior colleagues, technicians, pathology assistants, junior colleagues and artificial intelligence.
- Where to find information for various situations. It includes which person or which search engine is most useful for various clinical situations. See the Using resources section below for further information. Similarly, a pathologist often needs to be efficient at finding what is relevant among a large amount of information, and be able to integrate it into a decision or a relatively short report. Experience in looking up information will eventually give you an idea of where to locate specifics that you haven't even seen before. For questions where a look-up does not readily give the answer, a new trainee usually gets satisfactory replies even from senior trainees, but the more experienced you get, the more you need to establish contacts with particular people, such as certain subspecialties, who are most likely to be able to answer your questions when needed.
- How to distinguish memorization-worthy versus look-up versus practically useless information. With the ease of access to pathology information on the Internet through smartphones and computers, those studying for the everyday practice as a pathologist should not waste time memorizing what can essentially always be conveniently and timely looked up when needed. This includes most of the content of books and web pages that are sorted by names of diseases and conditions, because if the name of a disease is already known, then it can relatively quickly be looked up when needed in such sources. When finding relevant look-up information on the Internet or local repositories, it is generally enough to remember enough of its related words in order to find it again. After all, other doctors and even laypersons can look up diseases and conditions themselves, without the need for a pathologist consultation, so the expertise of memorizing such readily available information is expendable. Similarly, electronic systems are better suited than the human brain for the storage and rapid retrieval of databases of for example immunohistochemistry and genetics results of various diseases. The topics listed in this section are already immense enough to cover a lifetime of learning. Learning pathology therefore optimally involves the memorization of where and how to find look-up information, rather than directly memorizing such information. Therefore, if you need to choose a textbook to study, then choose one that you will always be able to quickly reach through your smartphone whenever you need its look-up information. Accordingly, the final aim of this resource is to help pathology trainees to give accurate and clinically useful reports to clinicians, in synergy with the resources that are available in everyday pathology practice, and to establish routines to continue such practices. The intended end result is to help clinicians to improve the lives as much as possible to as many people as possible, even if it means forgoing the prestige of becoming a walking encyclopedia. After all, life is not a Trivial Pursuit game; Don't fill your brain with random facts, even pathology-related ones. Rather, use high-yield learning sources, for even if they may take longer time to find, you will be better prepared for real life than when you study low-yield knowledge. For instance, the optimal studying is generally not of diseases individually, but rather of patterns of signs and other findings and the likelihoods of each differential diagnosis thereof. Arguably the best method of acquiring such knowledge is to be involved in many real cases at a pathology department, making your own diagnostic thinking of them, and correcting your thinking based on how you differed from a senior's decision on the case. Real life experience also gives you a better idea of what is memorization-worthy versus look-up information in various resources. After real life experience, the next best resource is arguably to solve virtual cases such as found in pathology qbanks.
As a general rule, the following factors makes a piece of information a look-up one:
- The probability of realizing that you don't know it in situations where it makes a difference. For example, when being presented with a numerical test result and you do not know the normal reference range for it, you at least realize that there are reference ranges out there that you can look up to help you. Thus, reference ranges are generally look-up information. It is generally the opposite for pitfalls. For example, if a person thinks a breast nodule looks like benign fibromatosis but does not know (or does not consider) that fibromatosis-like metaplastic carcinoma (FLMBC) looks similar, the probability is relatively low that the person will look that up.[note 1]
- The ease and speed of looking it up in reliable sources. Decisions with higher patient impact require more reliable sources. For example, the first forum answer from an anonymous user can be sufficient for an inconsequential decision, whereas information that may make a diagnosis benign versus malignant generally needs to be found on credible and peer reviewed sites.
- How to efficiently study for exams that contain questions that require direct memorization of look-up (or even practically useless) information, as long as such exams are in the way of practicing pathology. The best approach is arguably to spend enough time and effort on memorizing what is memorization-worthy, to the point where you will pass an exam even if you will fail multiple questions that require memorization of look-up or practically useless information. However, when starting pathology training, do not focus on studying for any final exams that are years away, because the content on such exams is systematically very different from what you need to know initially. For instance, you initially need to know how to handle the common types of cases, but exam makers on the other hand, in their effort to distinguish those who have studied more extensively than others, will systematically choose presentations that are not commonly encountered. Further information: Secrets Further exam advice is found in the Secrets chapter at patholines.org.
| Type of information | Examples (usually) | Learning approach |
|---|---|---|
| Memorization-worthy |
|
Directly memorizing it. |
| Look-up |
|
Knowing where to find it when needed, and how to use it. |
| Low-yield |
|
Instead studying patterns of findings and signs, and how they support various differential diagnoses. |
| Practically useless |
|
Avoiding, or skipping to potentially useful parts. |

Whether any piece of information is useful versus useless depend mainly on:
- The likelihood of a situation where the information is useful.
- The benefit versus detriment of having versus not having the information at hand in such a situation.
The clinical management aspects that can be useful for pathologists to learn are the thresholds for pathologic findings that substantially change the clinical management, such as a cancer stage that determines whether clinicians will try to completely remove all tumors versus treat palliatively. With such knowledge, pathologists can put relatively more effort on the cases and parts thereof that make the most difference to the patient. Otherwise, clinical management knowledge is generally useless to a pathologist.
Lectures may be memorization-worthy depending on the content, but a lecture of look-up-information is practically useless unless you maintain a system to quickly retrieve the information when needed. Thus, sometimes, it is better to study something more memorization-worthy while pretending to listen to a lecturer.
Also keep in mind that pathology in our look-up era is mainly a processing work rather than a memorization one, since your most important skills are to interpret findings, and sometimes involve a vast amount of information in order to suggest the most likely diagnoses. Again, arguably the best method fur such purposes is solving real or virtual cases.
Using resources
This resource is written with the intention to teach you what to do in the most common situations you are expected to encounter during your first years of pathology training, at least until the point that you are usually fairly confident about what disease or condition you have at hand, because then you know what words to use to look it up in the vast literature out there. At that point, the fastest way to get more information is generally by Googling the disease or condition name, followed by pathology outlines (which is generally the most likely to quickly give you the information you need), since Internet will always be readily available in pathology.[note 2] The most efficient method of studying to solve a case at hand is to not read articles in their entirety, but to scroll directly to the parts that are most likely to give you the information you need (generally past the first half of Pathology Outlines articles). A major skill of a pathologist is to have a good idea of what pages and texts can be skipped by only a quick glance.
Specific search methods for some specific purposes include the following (and the author has no financial or other conflicts of interests in mentioning any of these):
- Google, and then clicking the Images tab, if you just want to see more micrographs of the disease or condition. Even when you don't have a clue what condition you are looking at, you may find something that looks similar to your case by entering words you would use for the microscopic findings that you see.
- Adding cancer.net staging in Google searches for definitions of cancer stages, for example Googling prostate cancer cancer.net staging. The first search will then generally be the one from the American Society of Clinical Oncology.
- Googling "CAP synoptics" for guidelines for the most common cancer locations and types [1]. Don't google individual protocols, bypassing the overview list of protocols, as it may cause you to click on an outdated protocol.
- Considering a subscription for ImmunoQuery[note 3] (or equivalent database if you find one) in order to generate the most pertinent immunohistochemistry stains when you have two or more differential diagnoses for a case at hand. It is not sufficient to just memorize a few usually positive or negative immunostains for each disease and condition, because what you need in reality is to figure out what immunostains to order so that you can pinpoint one diagnosis among your multiple differentials. To master that, you will need to memorize a vast amount of immunostains and at what percentages they are positive for a vast amount of diseases and conditions, or you can have an online service like ImmunoQuery do that work for you. After all, in pathology there is essentially no immunohistochemistry that is so emergent that you do not have the time to use a computer or smartphone to look it up.
- ClinVar to look up the pathogenicity of specific genetic variants/mutations.
If the above resources do not provide a sufficient answer, more general search tools are mainly:
- Googling what you are looking for. You may add the word pathology or equivalent if results are too clinical or layman-oriented. Before even reading the title of results, first look at the URL. If it is from a reputable organization then you may proceed to check if the title is pertinent to what you are looking for, but if it is from an unfamiliar site then you should only proceed if you don't find a better source among your search results. On such unfamiliar sites, you will need to look for additional proof of reliability such as authorship of the content in order to use it as part of any diagnostic decisions.
- ChatGPT is useful in answering specific questions, or generating texts with particular content or format. However, don't make significant diagnostic decisions based on its answers without first verifying it with reliable sources, as it might give a false answer despite appearing confident.
You can generally rely on books and articles for presumably well-established topics (such as anatomy, and relatively common diseases). However, for potentially controversial and experimental topics, you generally need to ask yourself if the author has a conflict of interest before using its information for important decisions.
Ask a colleague at least whenever your own memory or a resource search is not enough, and there is a significant risk that you may do something irreversible that will negatively affect a patient.
Regarding lectures and knowledge your colleagues will tell you, make a judgment for each piece of information if it fits a criterion for memorization, or if it merits being written down somewhere you can find it so that you can look it up when needed.
Wikipedia often shows up among top Google results, and is generally accurate.[3]
Whenever possible, use textbooks and other sources that you can quickly access online, because you will likely always be close to the Internet, but not always close to your collections of physical material. Keep in mind that for the vast majority of content, you only need to have a hunch of where to find it again when needed, or what search terms to use. If something may be difficult to find again, you may add it to a personal digital collection to be readily available in times you need it.
Strive for sources that, taken together, are comprehensive enough for the presentation at hand. For example, if you first look in a resource on only benign conditions that may cause your presentation, you generally need to look for another source that includes malignant conditions as well.
Generally skip any part that tries to explain a microscopic appearance using only words, since a Google image search is generally more useful.
Test question
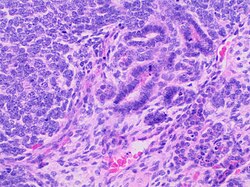
You are a new resident at a community hospital that is closely affiliated with a university hospital. The attending gives you a couple of slides from a case that is not yet signed out to preview and then come back with what you think would be the next steps. You are also told to share them with a med student who rotates at the department. One of the cases is from a kidney of a 4 year old girl. You look up the patient's history and she was admitted for orthopedic care for multiple fractures, and CT incidentally found a kidney tumor where workup was initiated as an inpatient. Microscopy shows the image at right. The med student has a smartphone and by googling "common kidney tumors in children", Wilms' tumor (also known as nephroblastoma) shows up as one of the top choices. You look up the condition on Pathology Outlines and the case seems to fit the microscopic description with its 3 typical components (second image at right).[4] On the same web page you also quickly see the following pieces of look-up information:
Which of the following would be the best next step if you were to decide yourself?
A major skill of a pathologist is having an idea of one’s unknowns. Furthermore, pediatric oncology is generally outside the limits of independent practice of any individual pathologist, generally requiring at least consultation with a colleague before diagnosis, classification and decisions about further workup (which in this case may be made at a pediatric oncology center). Thus, even if you though you knew how to proceed on your own, saying "I don't know" and asking for help is still the most correct as a new resident for a case like this.
You can skip reading these if you are already agree with the explanation above.
|
Using this resource
In this resource, it is recommended to read attentively until the end of the emergent pathology section. That part can be regarded as mainly describing concepts, which are relevant to read through and understand or at least consider. The rest will mainly provide guidelines for various situations, such as when you are presented with a biopsy of the gastrointestinal tract. Memorization-worthy information will generally be highlighted by: Memorization-worthy:[note 4], otherwise it is recommended to scroll quickly through such situation-based chapters, just to get an idea of what kind of information is available there, and then keeping this resource at a known location for whenever you are in that situation.
- Memorization-worthy:[note 5] This resource is available open-access and ad-free by googling patholines and then Starting pathology (handbook).
This resource is aimed at presenting the most important knowledge that you will need when starting pathology. For further knowledge that you will need as a pathologist and any subspecialization, the best way is to learn from reality, by solving the cases and problems that get presented to you, and thereby look specifically for the information that you will need among the immense literature and other information that is out there.
Also, this resource assumes that you use additional sources when needed (such as The WHO Classification of Tumors or others in the Using resources above). Therefore, it will not simply repeat such information, but may display external links to further information.
This resource also assumes that you will ask a colleague for advise if you see an unfamiliar presentation, and any literature lookup thereof still does not explain it. Therefore, this resource will focus on the most common presentations and the main differential diagnoses thereof, which may include rare conditions if the clinical management thereof would be significantly different. On the other hand, this resource omits rare conditions that rarely look similar to common presentations. Therefore, keep in mind that there are many such rare conditions that you do not know about, so keep asking colleagues whenever a presentation remains strange to you, and choose initial workplaces where you have experienced people to ask if needed.
Be tolerant to encountering many repetitions of the same content in related articles, since for example a cervical biopsy and a cervical cone have similar microscopic evaluation.
Using question banks
To test your knowledge on memorization-worthy information, seek questions that present you with a possible real life situation, as well as all pertinent information you can readily look up before needing to answer the question, so to ensure that it tests you on pertinent information as per the items earlier in this chapter. On the other hand, don't waste time on questions whose answer requires memorization of look-up or practically useless information (except if you want to practice looking up the answers online). There are about 5700 questions in PathPrimer, PathDojo, BoardVitals, and the ASCP Question Bank combined,[note 6] so you can be picky about which questions to take and which ones to skip.
Consider the usefulness of each question before spending significant time memorizing its answers or explanations, in order to maximize the useful knowledge you will learn overall. Generally, skip questions that test you on look-up information and practically useless information. Whenever you have a question where you can not picture in what real life situation it would be relevant to remember, then you can generally assume that it never will be. For questions that consists of a useful item together with a more useless one, only learn the the useful part and move on. If you can, flag/mark questions on memorization-worthy information that you did not know, and move on fairly quick on those as well, and later revisit those questions specifically if you have the time, as repetition is generally more efficient than lingering.
A common sign that a question will deal with look-up or practically useless information is where it asks which of the alternatives is false or correct for one particular condition, and the alternatives are relatively heterogenous (such as one dealing with diagnosis and another one dealing with prognosis). The high prevalence of such questions is mainly because they are easy and quick create, because the question creator can simply look up a condition, choose some more or less random facts, and then change small details to create alternatives. It requires no real life experience in everyday pathology practice, and learning from such a question is hence generally not useful for that purpose either.[note 7] The thought processes of real life pathology is generally the opposite of this, because you are generally presented with a specific location, gross appearance and microscopic appearance, and you need to practice on finding the most likely causative condition thereof among many differential diagnoses. Even other kinds of memorization-worthy pieces of information generally have a more limited scope among alternatives (like knowing the optimal staining duration in hematoxylin for a frozen section among alternative durations (see Emergent pathology). Thus, to practice your knowledge in memorization-worthy pathology, it is generally most efficient to skip false-versus-correct questions, in order to spend your limited time on something more worthwhile. Similarly, questions are in general rather low-yield whenever they deviate from the realistic direction.
Also, prefer succinct questions, that give you only the most relevant information in order to test the key learning point, without having to spend effort finding the most relevant information in long patient presentations, because you will get enough practice of that in everyday scrolling through patient charts.
If you have gone through all 5000 questions of the main question banks, and still have plenty of time for a second round, then you may work on lower yield questions as well. For example, you may practice on looking up answers for look-up questions, unless the presentation makes it clear that you cannot timely and conveniently do so (such as being in a rush during a frozen section).
Test question
You want to practice your memorization-worthy knowledge in real life pathology and not for exams (Further information: Secrets 'as per the Secrets chapter at patholines.org.). You find the following multiple choice question: "Which of the following statements for squamous cell carcinoma (SCC) of the skin is false?"
What is the most efficient way of dealing with this question for real-life pathology practice?
You can skip reading these if you are already agree with the explanation above.
|
Other indications for skipping a question include:
- Unrealistically withholding information, such as mentioning what kind of test identified the condition without giving you the test result (such as a case presentation that includes "Serology identified the organism").
- Unrealistic chronology, that is, not following the order of actual processes. It may include giving you the diagnosis and asking you what would be the expected clinical history.
- Showing you a molecular structure and asking you to identify the substance or its category, which is extremely unlikely to be a task in real life, and which can generally quickly be looked up (such as googling search by image and clicking the camera icon, followed by uploading the image of the structure, such as a photo or screenshot thereof).
- Unpractical statistics that is very unlikely to be used diagnostically, mainly because their calculations would be so complex that it is not worth it. For example, in reality you generally have a pattern of signs and other findings and you want to know the likelihood of each differential diagnosis, and for this purpose you only get limited help by knowing the reverse likelihood of what percentage each individual condition is to display the finding, because it would still be necessary to also know the epidemiology of each disease to calculate its likelihood.
Learning clinical pathology
For learning hematopathology, focus on the aspects of it that you may be expected to handle rather independently, mainly including peripheral blood smears and general screening of lymph nodes. Otherwise, a hematopathology workup is generally a very complex matter that should be performed by subspecialists thereof. In the unlikely event that you will end up at a workplace without an internal arrangement for how to consult a hematopathologist, make such arrangement yourself, since it will save you a lot of time compared to even trying to learn yourself how to perform a modern hematology workup.
For molecular pathology, focus on learning the situations in which molecular workup is needed, rather than the specific derangement. The latter can generally easily be looked up when needed, whereas the former can easily be missed.
Microbiology:
Put substantial effort on identifying what is actually memorization-worthy before starting to memorize microbiology-related content. For example, have an idea of the main workflows in a typical microbiology lab, but do not waste much time memorizing bench tests (negative/positive for indole, lactose/sucrose fermentation etc. etc.). In everyday practice there are only a limited number of bacteria that are identified by such testing, whereas the rest are generally identified by multitarget assays and/or MALDI-TOF. For exams where the passing score is an average of multiple topics (including the American Board of Pathology Certification Exams), focus on more useful topics instead.
General advice
- When making a mistake, admit that you did it and learn from it so as to focus on not repeating it. Also learn from the mistakes of others.
- Say "I don't know" instead of making up answers for what you do not know.
- Ask for help whenever needed, such as first time you are doing something, or whenever you are not sure about what to do, especially when doing something potentially irreversible. Also ask for help in moments whenever there is a high risk that you will not achieve what you need to do within a clinically acceptable time. Still, before asking, try to do as much as you can, as long as you do not do anything potentially irreversible, so that you can evaluate how you did it compared to the standard, and thereby know better how you will do it next time.
- Try to fit findings with the clinical picture so that the report makes sense, but do not make up findings that you do not see, especially in cases where you copy-paste words from a previous report or template. At the same time, do not omit relevant features just to fit an expected story.
- Do not wait for the whole pile. Whenever you can, do not be idle or do less urgent work while there is a pile of more urgent work gathering for you elsewhere. Instead, be familiar with where such piles are forming, and go there and grab whatever you may start working on right away.
- For larger specimens that need fixation before final grossing, you can still start writing a report of measurements and other externally visible findings to save time for later.
- Save your digital reports frequently.
- Focus on learning pitfalls and the interpretation of visual patterns and other non-written results, and do not waste much time memorizing information that can essentially always be conveniently and timely looked up when needed (but have an idea of where to find it). Further information: Learning pathology
Emergent pathology
- REDIRECT Emergency pathology
Fixation
Immersion
Within an hour after removal from the body,[6] tissue samples should generally be placed in vessels with enough fixative to allow them to lie freely in the solution.[7] The standard fixation fluid is generally 10% neutral buffered formalin, which is roughly equivalent to 4% formaldehyde.[8] The ratio of tissue:formalin should be 1:5[9] to 1:10[10].[10]
Duration
The duration depends on tissue thickness, where formalin will penetrate and fix the tissue at ~1 mm/hour.[11]
When not to use formalin
The main exceptions to using formalin are mainly:
- Intraoperative consultation.
- Suspected crystals, such as a tophus or other specimen suspicious for gout versus pseudogout. These should be sent in alcohol or dry, since formalin will dissolve the crystals.
- Suspected lymphoproliferative disorders, such as lymph nodes (or other lymphoid aggregates) with a suspicion of lymphoma, where samples are generally put in a special solution for flow cytometry.
- Need for genetic testing, such as some cases of products of conception.
- Cytology specimens, which are preferably sent fresh (such as in red top tubes) to be processed within a few hours. If processing may be after a few hours, put tubes on ice, or add 50% alcohol.[12]
- Need for microbiology evaluation, mainly bacterial culture. For potentially infectious workups, check with the microbiology lab if they have the tissue they need before putting the specimen in formalin.
- Need for immunofluorescence, such as immune complex-mediated disease, where specific preservation will give better test sensitivity.[13]
If you don't know, and if you cannot soon get in touch with anyone who can guide you, specimens can generally be stored in a fridge in the meantime, even overnight if it is late (but make sure to follow-up as soon as possible in the morning). Until then, don't put the specimen in formalin and don't freeze the specimen.
Gross processing
Following are general notes on selection and trimming in pathology.
Priority
|
1. More invasive intraoperative consultations |
For prioritizing when you have more than one thing ongoing at the same time, the list at right can be used.
Fresh specimens
The initial processing of fresh specimens is also termed triaging.
- For intraoperative consults including frozen sectioning, see separate article on Emergent pathology.
When triaging a specimen, the measurements are most important as these may change after fixation. Other descriptions can generally be made after the specimen is fixed.
- See also separate article on fixation, including what specimens should not immediately be put in formalin (mostly tophus, lymph nodes and products of conception).
Before cutting
- Confirm that the patient identity on the specimen container matches the identity that will be applied to the gross description and cassettes. {{If the referral or requisition form is available, confirm the patient identity on that one as well.}}
- (Check for any discrepancy between the specimen description on the container and on the referral or requisition form, such as left versus right.)
- Don't omit any piece in the container, such as ones hidden in wraps.
- Generally measure in 3 dimensions, or in volume, but the greatest dimension is generally enough for specimens less than 0.4 cm.
- Generally weigh entire organs, after having any attached tissue trimmed away if feasible.
- (Note the color of the sample, even when unremarkable, but do not linger on deciding it.)[note 8]
- Generally, use inking for resection margins where tumor radicality is important. Further information: inking
- (On fatty or greasy surfaces, apply vinegar to emulsify and remove the fat, dry the specimen and then ink. Otherwise, vinegar can be used either before or after inking to "dry" it.)
- (Preferably photograph or make a drawing where slices have been taken.)[14]
- Remove any surgical stitches from samples before microtomy.
- (At least for larger samples, consider looking for medical imaging or biopsy reports in order to better guide the process.)[15]
- Fix bone in formalin prior to decalcification. Use reminders so not to forget bone that is decalcifying.
- Plan in which order to cut different parts so as to be able to take all relevant sections. Generally sample relevant surgical margins and smaller parts first, as these may be harder to find or even be fragmented later on.
Cutting
- When cutting with the longer knives, try to cut in one stroke - do not use like a saw (continuous back and forth)
- Generally, strive to make slices perpendicular to visible interfaces of relevant tissues.
- Generelly dissect and inspect the entire specimen, while keeping relevant parts intact enough for presentation to seniors and/or maintaining orientation.
- Trim tissues for microscopy examination to a thickness of maximum 3-4 mm.[note 9]
Perpendicular versus en face sections
Two major types of sections in gross processing are perpendicular and en face sections:
- Perpendicular sections allow for measurement of the distance between a lesion and the surgical margin.
- En face means that the section is tangential to the region of interest (such as a lesion) of a specimen. It does not in itself specify whether subsequent microtomy of the slice should be performed on the peripheral or proximal surface of the slice (the peripheral surface of an en face section is closer to being the true margin, whereas the proximal surface generally displays more area and therefore generally has greater sensitivity in showing pathology, also compared to perpendicular sections).
- A shaved section is a superficial en face slice that contains the entire surface of the segment.
Tissue selection
When sampling sections to submit for microscopic examination, whenever you sample from something that looks abnormal, generally try to also sample from the same type of tissue that looks normal.[note 10]
Biopsy wraps, bags and sponges
Put the following types of specimens in bags:
- Tiny specimens that need to be poured out from their containers.
- Bloody specimens such as endometrial curettages or products of conception. For products of conception, chorionic villi may otherwise contaminate other specimens. Bloody specimens may stick to wraps, so generally avoid that situation.
- Friable tissue such as urinary bladder biopsies.
Put the following types of specimens in bags, wraps or sponges:
- Other tiny specimens
- ((Any small piece of tissue where there is no leftover specimen to retake sections, since tissues occasionally get lost from cassettes, and the absence of a wrap, sponge or bag in the cassette of such cases points towards a mistake made at gross processing.))
Specimens must be fixed enough to be put on sponges.
H&E staining urgency
(Even in departments where other staff are primarily responsible for determining the urgency of H&E staining of each specimen, still double-check that it is correct if you can, such as by cassette color.) A major indication for rushing cases through H&E staining is a high risk of cancer, especially where immunohistochemistry staining will likely be performed, and the decision and types of staining will be determined by the standard H&E stain. Tissues that are generally rushed are:
- Brain biopsy.
- Lung biopsy.
- Breast needle biopsy.
- Biopsy from known tumor tissue.
- Suspected malignant lymph nodes, including lymphoma. However, these are generally not urgent when submitted together with a tumor, except mainly for the following (which are generally urgent):
- Pelvic sentinel lymph nodes
- Sentinel lymph nodes from known invasive lobular carcinoma (but not invasive ductal carcinoma)
In both these cases, the cases are rushed so that immunohistochemistry can be performed if a metastasis is not readily detected on standard H&E slides, so that it is available by the time the rest of the slides are out. Immunohistochemistry in these cases can detect micrometastases that are not readily visible on H&E stain, but are evident on cytokeratin AE1/AE3. However, if the lab stains such cases regardless of whether H&E stain shows a metastasis or not, then they do not need to be rushed.
Marking cassettes
Use only hard pencil (or specially purchased histology markers), as marks made with pens, Sharpie markers, or scientific freezer-safe markers can get dissolved in tissue processing.[16]
Evaluation of tumors
- REDIRECT Evaluation of suspected malignancies
Reporting
Following are general notes on reporting in pathology.
- Save your digital work frequently, and also before you leave a computer, even if you think you'll be back shortly. If you have many small specimens to write up in the same report, you may want to save every 2 to 3 specimens. It doesn't matter how much time and effort you spend on something if you're just going to let it disappear in the next glitch.
- Double-check your report, especially if you copy-pasted and adapted a previous report rather than using a template with blank fields or making your report from scratch.
Components
Selection and trimming
From the stage of selection and trimming, a histopathology report should preferably include:
- Case:
- Patient identification and/or sample number
- Type of tissue sample as described on container
Microscopic evaluation
- Specimen chronology, often A, B, C, etc., at least where there are multiple specimens from the same case. With multiple specimens, preferably write out the chronology for all of them first, so that you don't miss reporting any of them later.
- Specimen type and/or surgery type, such as "appendix, appendectomy", for clarification. This is not necessary at all departments. For the procedure, use the same term as the operating report whenever possible.
- Microscopic description. This is not always necessary, but should be included if the diagnosis is uncertain. One systematic approach is to describe findings from largest to smallest ones. For example, a description of a tumor can start with the demarcation of the tumor, followed by texture, cell shapes, nucleus shapes and chromatin appearance.
- Diagnosis or most probable diagnoses.
- If the diagnosis does not clearly account for all conditions that were requested, suspected or asked to be ruled out by the referring clinician (such as stated on the requisition form), you need to classify the specimen as "positive for" versus "negative for" for each such condition, or give a reason for why an evaluation thereof could not be made.
- In case of malignancy or suspected malignancy:
- Depth or most distant invasion of malignant findings.[17] Depending on location, it may need to exclude important pathways, such as vascular, neural and/or through capsules or other layers.
- Whether the resection is radical or not.
Depth
Factors supporting a relatively more comprehensive report, particularly in the inclusion of negated findings:
- Lack of explanation from existing evidence. For example, an inflamed appendix that fits the medical history does not need detailed mention of harmless incidental findings.
- Prospective review: If your report is likely to undergo double-reading by another pathologist before sign-out, it should either be more detailed, because the doctor who will do the double-reading then gets an idea of your thought process, including what you have looked for versus what may still need to be evaluated. If you know who will do the prospective review for a report of yours, you may alternatively convey your thought process by other means such as directly talking to that person.
- Highly suspected locations, such as given from the referral.
- Difficulty in obtaining the specimen, such as a CT-guided biopsy versus a skin shave.
- Defensive precautions, which appears to be more common among doctors in the Unites States compared to for example Europe.[18][19]
Multiple instances of the same type of pathology (such as lung nodules) can often simply be reported as such, at least with a particular mention of the largest or the most severe example thereof.
Uncertainty
|
| probably likely |
| suggesting |
| suspicious for |
| possibly |
| (benign condition) cannot be excluded |
| not likely |
| (malignant condition) cannot be excluded |
|
When something looks very much like a specific entity but you are not sure, preferably use "-like" (or when feasible, "-oid" such as squamoid for squamous-like cells).
When the clinical picture strongly favors a certain condition, and the pathology favors it as well, findings are generally described as "consistent with". Sometimes, "bordering on" can be described when the picture almost fits specified criteria of a specific diagnosis.
It is alright to consider the diagnosis of a pathology report to be a combination of the clinical picture and what can be seen on the specimen. For example, if the microscopy picture is uncertain, you may to a certain degree tend towards the diagnosis that best fits the clinical picture. However, mention differential diagnoses if they are still significantly possible, and would confer a different treatment or another substantially different consequence.
For both findings and diagnoses, is preferable to write "negative for..." rather than "no..." to emphasize the possibility of false negative findings.
Synoptic reports
For cancers, generally include a synoptic report, such as per College of American Pathologists (CAP) protocols at cap.org/protocols-and-guidelines. However, synoptic reports are generally not needed for tumor metastases.
Sizes
Whenever possible, give numerical quantities of sizes, rather than descriptions that are subjective (such as "small" or "large") or variable (such as "apple-sized").
Tailoring
The information contained in the reporting sections in this resource assume that the clinician has requested the exam for the topic at hand, but should still be tailored to any particular questions or requests by the clinician. Any relevant findings beyond the issues or questions raised by the clinician should also be mentioned.
As there are generally local practices about how to write reports, it is recommended to have either a personal or practice-specific repository of report templates that you can copy-paste for various cases. When doing so, however, use marks or empty spaces for relevant findings that are frequently changed in the template. Such marks should be conspicuous, so that the report clearly looks unfinished if you haven't tailored it to the case at hand (such as "...measuring _____."). Thereby, you avoid omissions or even wrongly entered information from templates.
The most important findings are preferably moved to near the top of the report if feasible.
Example of report format:
A. Organ, side/site, procedure:
|
Wording
If a certain grammatical rule has a risk of making the report less clear to the reader, ignore that rule in that situation.
Restrict acronyms/abbreviations to those who are certainly well known among all doctors, such as "cm".[note 11]
Generally describe what can be seen rather than processes (such as preferring "an abundance of" rather than "proliferation of").
If using a dictation device, avoid "no", and instead use "negative for" (and "positive for" in opposite cases), since there's a risk of "no" not being transcribed and thereby creating the opposite meaning.
Avoid "evidence of" unless you feel comfortable with the legal implications of its meaning in the report.
Whenever there is text needing formatting (text size, font type and/or UPPER vs lower case), it is generally most efficient to do it all at once after all the text is written.
Skin excisions
In skin cancers, use "peripheral" or "radial" margins (whereas "lateral" margin should be reserved for the margin opposite to the medial margin).[20]
Notes
References
- ↑ . Copyrightability of Tables, Charts and Graphs. Deep Blue Repositories, University of Michigan Library. Retrieved on 2022-01-07.
- ↑ Image by Mikael Häggström, MD, using source images by various authors. Source for useful context in problem-based learning: Mark A Albanese, Laura C Dast (2013-10-22). Understanding Medical Education - Problem-based learning. Wiley Online Library.
- ↑ Kräenbring, Jona; Monzon Penza, Tika; Gutmann, Joanna; Muehlich, Susanne; Zolk, Oliver; Wojnowski, Leszek; Maas, Renke; Engelhardt, Stefan; et al. (September 24, 2014). "Accuracy and Completeness of Drug Information in Wikipedia: A Comparison with Standard Textbooks of Pharmacology ". PLOS ONE 9 (9): e106930. doi:. PMID 25250889. Bibcode: 2014PLoSO...9j6930K.
- ↑ Ellen D’Hooghe, M.D., Gordan M. Vujanic, M.D., Ph.D.. Kidney tumor - Childhood tumors - Nephroblastoma. Pathology Outlines. Topic Completed: 16 September 2020. Minor changes: 6 October 2021
- ↑ Shaofeng Yan, M.D., Ph.D.. Skin nonmelanocytic tumor, Carcinoma (nonadnexal), Squamous cell carcinoma. Pathology Outlines. Topic Completed: 13 January 2020. Minor changes: 8 December 2021
- ↑ . Breast pathology grossing guidelines. UCLA Health. Retrieved on 2021-09-09.
- ↑ Katarzyna Lundmark, Krynitz, Ismini Vassilaki, Lena Mölne, Annika Ternesten Bratel. Handläggning av hudprover – provtagningsanvisningar, utskärningsprinciper och snittning (Handling of skin samples - Instructions for sampling, cutting and incision. KVAST (Swedish Society of Pathology). Retrieved on 2019-09-09.
- ↑ . Paraformaldehyde, Formadehyde and Formalin. Duke University. Retrieved on 2019-12-17.
- ↑ . Fixation of Tissues. Approval Date: August 2016, August 2020. Review Date: August 2024|website=Royal College of Pathologists of Australia
- ↑ 10.0 10.1 Buesa RJ, Peshkov MV (2012). "How much formalin is enough to fix tissues? ". Ann Diagn Pathol 16 (3): 202-9. doi:. PMID 22483550. Archived from the original. .
- ↑ . How to Submit Tissues for Embedding. University of Pittsburgh, Starzl Transplantation Institute. Revised 04/19/21
- ↑ . How to send fluid and make good cytology slides. Tufts University.
- ↑ Mubarak M, Kazi Javed I, Kulsoom U, Ishaque M (2012). "Detection of immunoglobulins and complement components in formalin fixed and paraffin embedded renal biopsy material by immunoflourescence technique. ". J Nephropathol 1 (2): 91-100. doi:. PMID 24475396. PMC: 3886135. Archived from the original. .
- ↑ Monika Roychowdhury. Grossing (histologic sampling) of breast lesions. Pathologyoutlines.com. Topic Completed: 1 August 2012. Revised: 19 September 2019
- ↑ . Gross Pathology Manual By The University of Chicago Department of Pathology. Updated 2-14-19 NAC.
- ↑ . Histopathology Services. UNC School of Medicine. Retrieved on 2021-11-15.
- ↑ 17.0 17.1 17.2 17.3 . An Example of a Melanoma Pathology Report. Melanoma Foundation. Retrieved on 2019-09-24.
- ↑ Studdert D. M.; Mello M. M.; Sage W. M.; DesRoches C. M.; Peugh J.; Zapert K.; Brennan T. A. (2005). "Defensive medicine among high-risk specialist physicians in a volatile malpractice environment ". JAMA 293 (21): 2609–2617. doi:. PMID 15928282.
- ↑ Steurer J.; Held U.; Schmidt M.; Gigerenzer G.; Tag B.; Bachmann L. M. (2009). "Legal concerns trigger PSA testing ". Journal of Evaluation in Clinical Practice 15 (2): 390–392. doi:. PMID 19335502.
- ↑ David Slater, Paul Barrett. Standards and datasets for reporting cancers - Dataset for histopathological reporting of primary cutaneous basal cell carcinoma. The Royal College of Pathologists. February 2019
Cite error: <ref> tags exist for a group named "note", but no corresponding <references group="note"/> tag was found, or a closing </ref> is missing