Peripheral blood smear
Author:
Mikael Häggström [note 1]
Look at and comment separately on white blood cells, red blood cells and platelets. You generally don't need to aim for a perfect report, because for most purposes, a peripheral smear is a relatively simple screening test, and clinicians may opt to perform for example flow cytometry and/or a bone marrow biopsy if they want a better evaluation.
Contents
Comprehensiveness
On this resource, the following formatting is used for comprehensiveness:
- Minimal depth
- (Moderate depth)
- ((Comprehensive))
- Other legend
<< Decision needed between alternatives separated by / signs >>
{{Common findings / In case of findings}}
[[Comments]]
Link to another page
Oil immersion microscopy
This is preferred for light microscopy of peripheral blood smears in order to achieve a very high magnification. First use low or medium power to center on suspicious cells, or where red or white blood cells are best appreciated. Put a drop of immersion oil on the location and switch to the immersion objective. Then, whenever there's oil on a slide, always think twice before switching between objectives so as to avoid getting oil on any of your dry objectives (which is a bit tedious to clean off).
Red blood cells
Automated values
When available, automatic quantification of mean corpuscular volume (MCV) and red blood cell (RBC) distribution width (RDW), usually as part of CBC panel, generally decides whether you will call the sample "normocytic" versus "microcytic"/"macrocytic" and/or "anisocytotic", even if it is not clearly visible in the microscope. If automated values are not available, compare RBC sizes to lymphocyte nuclei, which should normally be the same size. If mean corpuscular hemoglobin (MCH) and mean corpuscular hemoglobin concentration (MCHC) are normal, but you still see multiple RBCs with central pallor greater than 50% of the diameter, you can report it as "Increased central pallor", and you may add "indicating iron deficiency" if it is compatible with the clinical history.
Automated values can be graded as follows:[1]
| Interpretation | Mild | Moderate | Marked |
|---|---|---|---|
| Microcytosis | MCV : 70 - 79 | MCV : 60 - 69 | MCV <60 |
| Macrocytosis | MCV : 100 - 115 | MCV : 115 - 125 | MCV >125 |
| Hypochromasia | MCH : 23 - 26 | MCH : 21 - 23 | MCH <20 |
| Anisocytosis | RDW: 14.5[2] or 16[1] - 18 | RDW : 18 - 22[1] or 26[2] | RDW > 22[1] or 26[2] |
Morphologic findings
Look for poikilocytosis (red blood cells of abnormal shapes). These are counted as a percentage of visible red blood cells:[1]
- Burr cells versus spur cells
Burr cells are distinguished from spur cells by having more equally distributed and rounded projections. They are usually artifactual. However, they may also be caused by renal insufficiency, so if this is present, a report may include "Occasional/Multiple echinocytes, consistent with renal insufficiency".
- Intraerythrocytic findings
Platelets
If CBC is performed, use count to determine whether platelets are "normal in number" or whether there is "thrombocytopenia" or "thrombocytosis". If no CBC, count platelets within a high power oil immersed field, which should normally be 8 to 20.
Large platelets are those with a diameter greater than 4 microns. Giant platelets are those with a diameter greater than 7 microns (larger than a normal red blood cell).[3] Example report:
| Numerous large and giant platelets(, suggesting an increased platelet turnover)(( such as in immune thrombocytopenic purpura. They may also be present in myeloproliferative neoplasms, myelodysplasia, and some congenital thrombocytopenia syndromes, including Bernard-Soulier syndrome and MYH9-related disorders.[3])) |
In thrombocytopenia from automatic counting, look in particular for:
- Clumping of platelets (which can cause a falsely low automatic platelet count). If present, check with the lab if it was sent in EDTA (which may cause artefactual clumping) and ask to have a blood sample sent in sodium citrate instead. Also, look for satellitosis (platelets attached around white blood cells).
- Schistocytes among red blood cells.
White blood cells
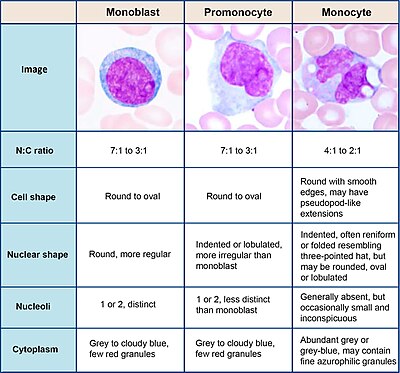
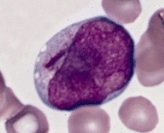
Look for:
Blast cells, generally having large nucleus, immature chromatin, a prominent nucleolus, scant cytoplasm and few or no cytoplasmic granules. This example has an Auer rod (to the left of the nucleus). Further information: Suspected blasts on peripheral blood smear
Hypersegmented neutrophils. This is abnormal when more than half of neutrophils have at least 4 segments, or more than 5% of neutrophils have more than 5 segments.[4]
In patients with known chronic lymphocytic leukemia, estimate the percentage of prolymphocytes, which are medium-sized lymphocytes with prominent nucleoli.[5] A percentage of less than 5% can be reported as such.
When smudge cells constitute more than 10% of white blood cells, or in patients with CLL, a separate smear with a drop of serum albumin to every four or five drops of blood should be made to stabilize cell membranes.[6] A semi-quantification of different cell lines should then be made on the albumin slide, whereas white and red blood cell morphology should still be made on the original slide, since it is altered by albumin.
Report
Example report:
| Normochromic normocytic red blood cells. Red blood cells show <normal morphology / anisopoikilocytosis with occasional ___>. [[If thrombocytopenia, also add "Schistocytes are not significantly increased" if applicable.]]
{{Leukocytosis with neutrophilia / lymphocytosis.}} White blood cells show no left shift or blasts. Platelets show no evidence of clumping, and show normal granularity. (Causes of the above findings include ___.) |
Notes
- ↑ For a full list of contributors, see article history. Creators of images are attributed at the image description pages, seen by clicking on the images. See Patholines:Authorship for details.
Main page
References
- ↑ 1.0 1.1 1.2 1.3 1.4 Unless otherwise specified in table, reference is:
- . Hong Kong Medical Technology Association - Quality Assurance Programme - Haematology and Serology, Prepared by HKMTAQAP Haematology & Serology Panel on November 2002.. - ↑ 2.0 2.1 2.2 . High RDW level in the blood. MrLabTest. Last update: 12/01/2021
- ↑ 3.0 3.1 Teresa Scordino (2016-12-02). Giant platelets. American Society of Hematology.
- ↑ Glassy, Eric (1998). Color atlas of hematology : an illustrated field guide based on proficiency testing . Northfield, Ill: College of American Patholgists. ISBN 978-0-930304-66-9. OCLC 40976106.
- ↑ . prolymphocytes in PLL. American Society of Hematology (2013-07-16).
- ↑ Fredrick L. Kiechle. Q & A. CAP Today. June 2010
Image sources