Renal tumor
Jump to navigation
Jump to search
Author:
Mikael Häggström [note 1]
Contents
Presentations
Total or partial nephrectomy.
Gross processing
For orientation, the ureter stump points to the inferior pole.[1]
Components:[1]
- Note the appearance of the renal surface
- Inspect the capsule and pericapsular soft tissue, and possibly an accompanying adrenal gland
- Measure kidney dimensions (and optionally weight)
- Identify the surgical margin of the ureteral stump, renal vein and renal artery
- In cases of a bulging tumor, apply ink to the surface of pericapsular fatty tissue by the tumor.
- Dissect the ureteral stump
- Split the kidney in the middle, in the coronal plane. Release the fibrous capsule. Dissect the renal pelvis and calyces. Inspect the parenchyma on cut surfaces.
- Note and possibly measure the medullary thickness and the width of the renal pelvis
- Note the appearance of any concretions
- Note the location and size of the tumor, macroscopic appearance of cut surfaces, delimitation compared to surrounding structures, and relation to the fibrous capsule and perinephric tissues. If the fibrous capsule can be detached from the tumor surface, the inside of the capsule is examined where it was in contact with tumor, in order to detect any tumor overgrowth or breakthrough.
Slices for submission
Including:[1]
- At least 3-4 pieces from the tumor, showing relationship to surrounding kidney tissue, renal capsule, perinephric tissues, renal pelvis, hilar adipose tissue and blood vessels.
- Part of the fibrous capsule if being in contact with the tumor (oriented perpendicular to the capsular surface)
- Margins of ureteral stump, renal vein and renal artery, and from hilus tissue (to detect possible tumor invasion of smaller blood vessels)
- One slice of normal kidney tissue
- Any detected lymph nodes in perihilar region
- At least one slice from any included adrenal gland.
- In case of cancer in the renal pelvis, at least three slices from the tumor-suspected area, including radicality slices from surrounding normal mucosa, and adjacent peripelvic fatty tissue. Take about 1 piece per cm2 from other parts of the renal pelvis, as well as a piece from the surgical edge of the ureteral stump.
Microscopic evaluation
Main components are:
- Determining the histologic type
- Staging
Histologic type
Look for the most common tumors:
Renal cell carcinomas frequently display hemosiderin and small cytoplasmic vacuoles as shown.[2]
| Renal carcinoma subtype | Characteristic morphology[3] | Positive immunohistochemistry[3] | Image |
|---|---|---|---|
| Clear cell renal cell carcinoma | Clear cells in nests or solid pattern, delicate branching fibrovascular network. | Vimentin, EMA, keratin, CD10, Pax2, RCC, CAIX (circumferential membranous) | 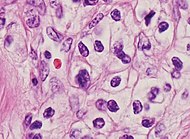
|
| Papillary renal cell carcinoma | Papillary architecture, small-to-medium-sized cells. Often foamy macrophages in fibrovascular cores. | Vimentin, keratins, CK7, AMACR, RCC | 
|
| Clear-cell papillary renal cell carcinoma | Clear cells in papillary, solid, or nested pattern, abluminal linear arrangement of nuclei. | CA-IX (basolateral membranous), HMWCK | 
|
| Chromophobe renal cell carcinoma | Distinct (vegetable-like) cell borders, raisin-like nuclei with perinuclear halos. | Ksp Cadherin, CD117, EMA, keratins, CK7 | 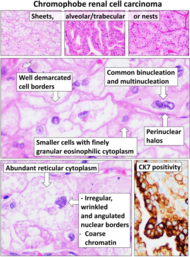
|
| Renal cell carcinoma (unclassified/without further specification) | Contains features not fitting in subtypes. It includes sarcomatoid tumors without recognizable epithelial elements, mucin-producing tumors, and mixtures of epithelial and stromal elements. | ||
| Oncocytoma | Small nests of cells, uniform small nuclei. | Ksp Cadherin, CD117, EMA, keratins | 
|
| Angiomyolipoma | Classically triphasic with myoid spindle cells, mature adipose tissue (seen in > 90% tumors) and dysmorphic thick walled blood vessels without elastic lamina. | HMB-45 (100%), Melan-A (87%) SMA (73%)[4] | |
| Multilocular cyst | Encapsulated, non-infiltrating and composed of non-comumnicating cysts filled with clear, straw-colored, or blood-tinged fluid.[5] | ||
| Cystic nephroma | Cysts lined by simple epithelium with "hobnail morphology", that is, the nuclei of the epithelium bulges into the lumen of the cysts. Ovarian-like stroma with pindle cell morphology and basophilic cytoplasm. |

| |
| Collecting duct carcinoma | Infiltrating high-grade adenocarcinoma centered in the medulla, desmoplastic stroma. | EMA, CK7, HMWCK, Pax2, Pax8 | |
| Microphthalmia Transcription Factor (MITF)- family translocation renal cell carcinoma | Clear and eosinophilic cells, voluminous cytoplasm, psammomatous calcifications (TFE3) Biphasic tumors (TFEB). |
HMB-45, Melan A, Cathepsin K, TFE3/TFEB | |
| Fumarate hydratase-deficient renal cell carcinoma | Variable, intracystic papillary pattern with prominent hyalinization and usually tubulocystic patterns. | Loss of FH (deficient), 2SC | |
| Succinate dehydrogenase-deficient renal cell carcinoma | Oncocytic cells with cytoplasmic vacuoles and inclusion-like spaces, neuroendocrine-like chromatin. | SDHB loss (deficient) | |
| Renal medullary carcinoma | High-grade adenocarcinoma, inflammatory, myxoid, or desmoplastic stroma, evidence of sickle cell disease/trait. | INI-I loss, Oct 3/4 positive |
Multifocality of tumors favors mainly papillary renal cell carcinoma (although most of these are unifocal).[6]
| Renal cell carcinoma type | RCC | CK7 | CAIX |
|---|---|---|---|
| Clear cell | 62% | 16% | 86% (box-like) |
| Clear cell papillary | 18% | 94% | 97% (cup-like) |
| Papillary | 81% | 74% | 61% |
| Chromophobe | 6% | 89% | 22% |
Staging
| Stage | TNM | Description |
|---|---|---|
| Tx, N0, M0 | Tumor cannot be assessed | |
| T0, N0, M0 | No evidence of primary tumor | |
| I | T1, N0, M0 | Tumor ≤7 cm; limited to kidney |
| T1a, N0, M0 | Tumor ≤4 cm; limited to kidney | |
| T1b, N0, M0 | Tumor 4-≤7 cm; limited to kidney | |
| II | T2, N0, M0 | Tumor >7 cm; limited to kidney |
| T2a, N0, M0 | Tumor 7-≤10 cm; limited to kidney | |
| T2b, N0, M0 | Tumor >10 cm; limited to kidney | |
| III | T3, N0, M0 | Tumor extends to major veins or perinephric tissue but not into ipsilateral adrenal gland nor beyond Gerota's fascia |
| T3a, N0, M0 | Tumor grossly extends into renal vein or its segmental branches, or tumor invades the pelvicalyceal system, or tumor invades perirenal and/or renal sinus fat but not beyond Gerota's fascia | |
| T3b, N0, M0 | Tumor grossly extends into vena cava below the diaphragm | |
| T3c, N0, M0 | Tumor grossly extends into vena cava above the diaphragm or invades the wall of the vena cava | |
| T1-T3, N1, M0 | The main tumor can be any size and may be outside the kidney, but it has not spread beyond Gerota’s fascia. The cancer has spread to regional lymph nodes (N1) but has not spread to distant lymph nodes or other organs (M0). | |
| IV | T4, any N, M0 | Tumor invades beyond Gerota's fascia |
| Any T, any N, M1 | Tumor has spread to distant lymph nodes and/or other organs. |
Report
The report should include:
- Histologic diagnosis
- Greatest dimension (from grossing)
- Grade
- Radicality
Example:
Kidney, left, partial nephrectomy:
|
Renal cell carcinomas should also have a synoptic report including:[1]
- Total or partial nephrectomy
- Tumor characteristics:
- In nephrectomy: Location
- Size
- Relationship to capsule, perinephric tissues, renal pelvis, adrenal gland and hilar blood vessels
- Histological type
- Degree of differentiation
- Radicality
- Any lymph node metastases
Notes
- ↑ For a full list of contributors, see article history. Creators of images are attributed at the image description pages, seen by clicking on the images. See Patholines:Authorship for details.
Main page
References
- ↑ 1.0 1.1 1.2 1.3 Monica Dahlgren, Janne Malina, Anna Måsbäck, Otto Ljungberg. Stora utskärningen. KVAST (Swedish Society of Pathology). Retrieved on 2019-09-26.
- ↑ . Kidney tumor - Adult renal cell carcinoma - common - Renal cell carcinoma overview. PathologyOutlines. Topic Completed: 1 April 2017. Minor changes: 15 May 2021
- ↑ 3.0 3.1 Unless otherwise specified, reference in table is: Sirohi, Deepika; Smith, Steven C.; Agarwal, Neeraj; Maughan, Benjamin L. (2018). "Unclassified renal cell carcinoma: diagnostic difficulties and treatment modalities ". Research and Reports in Urology Volume 10: 205–217. doi:. ISSN 2253-2447.
- ↑ Esheba, Ghada El Sayed; Esheba, Noha El Sayed (2013). "Angiomyolipoma of the kidney: Clinicopathological and immunohistochemical study ". Journal of the Egyptian National Cancer Institute 25 (3): 125–134. doi:. ISSN 11100362.
- ↑ Banner, MP; Pollack, HM; Chatten, J; Witzleben, C (1981). "Multilocular renal cysts: radiologic-pathologic correlation ". American Journal of Roentgenology 136 (2): 239–247. doi:. ISSN 0361-803X.
- ↑ Error on call to Template:cite web: Parameters url and title must be specifiedNicole K. Andeen, M.D., Maria Tretiakova, M.D., Ph.D.. . Pathology Outlines. Last author update: 1 March 2018. Last staff update: 28 September 2022 (update in progress)}}
- ↑ . Immunoquery. Retrieved on 2023-05-17.
- ↑ . Kidney Cancer Stages. cancer.org. Last Revised: February 1, 2020
- ↑ Swami, Umang; Nussenzveig, Roberto H.; Haaland, Benjamin; Agarwal, Neeraj (2019). "Revisiting AJCC TNM staging for renal cell carcinoma: quest for improvement ". Annals of Translational Medicine 7 (S1): S18–S18. doi:. ISSN 23055839.
Image sources

