Ductal carcinoma in situ
Author:
Mikael Häggström [note 1]
Ductal carcinoma in situ (DCIS):
Contents
Comprehensiveness
On this resource, the following formatting is used for comprehensiveness:
- Minimal depth
- (Moderate depth)
- ((Comprehensive))
Gross examination
As per:
or mastectomy.
Microscopic evaluation
Malignant epithelial cells confined to the ductal system of the breast.[1] The cells are cohesive and have high grade atypia.[2]
Differential diagnoses
- Invasive ductal carcinoma
- Has invasion through the basement membrane of over 1 mm in size.[3]
DCIS with microinvasion, defined as DCIS with focus of invasive cancer measuring up to 1.0 mm in size.[4]
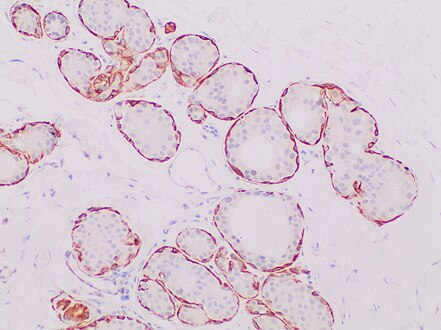
In uncertain cases, use immunohistochemistry stain for calponin (has the highest sensitivity) and p63 (has the highest specificity).
Immunohistochemistry for the myoepithelial marker[note 2] calponin in ductal carcinoma in situ, highlighting myoepithelial cells around all tumor cells, thereby ruling out invasive ductal carcinoma.
Invasive ductal carcinoma with tubular features can look like benign tubules, but calponin and p63 shows no surrounding myoepithelial cells.
There is no single definite cutoff, but the following are suggested cutoffs defining a ductal carcinoma in situ:[5]
- Size over 2 mm.
- Involving more than one duct.
- Lobular carcinoma in situ (LCIS)
Lobular carcinoma in situ (LCIS) displays discohesive cells, often a feathery clear space between cells, solid growth pattern, intracytoplasmic vacuoles, and lack of polarization around luminal spaces.[6]
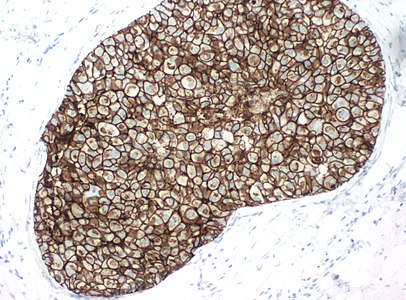
LCIS typically fills smaller lobules rather than ducts, but DCIS can display lobular cancerization as shown at bottom of image.[image 1]
When unsure, perform immunohistochemistry for E-cadherin and p120. Both E-cadherin (left image below) and p120 (right) have a membranous staining pattern in ductal carcinoma in situ:
In contrast:
Grading
At least a low/intermediate/high grading (by Van Nuys criteria) as follows:[7]
- Low grade DCIS
- Nuclei 10-15 microns (2-3 times the size of a red blood cell)
- Nuclei oval, round, regular, evenly dispersed chromatin up to mildly irregular and minimally pleomorphic
- Nucleoi, if present, are small and indistinct
- Intermediate grade DCIS
- Same nuclear features as low grade

- Substantial tumor cell (comedo) necrosis is present
- High grade DCIS
- Nuclei >15 microns (over 2.5[10] times the size of a red blood cell)
- Nuclei are pleomorphic with clumped chromatin
- Nucleoli are prominent, enlarged
- Necrosis is almost universal and lumenal
(Numerical grading
Use the low/intermediate/high grade to give a numerical grading as follows:[11]
| Feature | Points | ||
|---|---|---|---|
| 1 | 2 | 3 | |
| Nuclear grade | Low | Intermediate | High |
| Glands/papillae | >75% | 10% - 75% | <10% |
| Mitotic rate (per 10 HPF) | <1 | 1 - 2 | >2 |
| Central necrosis | <10% | 10% - 50% | >50% |
The points for each feature are added together, giving the following result:[11]
- 4 - 7 points: Grade 1
- 8 - 9 points: Grade 2
- 10 - 12 points: Grade 3
)
Extent
- In biopsies, do not state the extent of DCIS.
- In excisions, state the distance to the closest margin. (Additional extent specification is optional. If there is invasive tumor in addition to DCIS, you may give the DCIS extent in terms of the number of blocks containing DCIS. If an excision has DCIS without any invasive component, consider stating the total size in terms of the greatest distance between any DCIS foci.)
Microcalcifications

If invasive ductal carcinoma is seen, make at least a low power screening for microcalcifications (to correlate with imaging), but there's no need to look carefully (as tiny microcalcifications would unlikely correlate with imaging anyways).
Estrogen and progesterone receptors
Generally perform immunohistochemistry for estrogen and progesterone receptors and calculate the percentage of positive tumor cells.
HER2
((HER2 testing is not necessary, but can be done for prognostic profiling.[12])) See invasive ductal carcinoma for how to evaluate HER2.
Reporting

Example:
| Left breast mass, 2:00, 1 cm from nipple, ultrasound-guided vacuum assisted core needle biopsy: Ductal carcinoma in situ. Negative for invasive carcinoma. |
For cancers, generally include a synoptic report, such as per College of American Pathologists (CAP) protocols at cap.org/protocols-and-guidelines.
See also: General notes on reporting
Notes
- ↑ For a full list of contributors, see article history. Creators of images are attributed at the image description pages, seen by clicking on the images. See Patholines:Authorship for details.
- ↑ For myoepithelial markers, a combination of p63 and calponin is generally recommended for breast lesions. D2-40 is useful for highlighting lymphatics for invasion.
Main page
References
- ↑ Siziopikou, Kalliopi P. (2013). "Ductal Carcinoma In Situ of the Breast: Current Concepts and Future Directions ". Archives of Pathology & Laboratory Medicine 137 (4): 462–466. doi:. ISSN 0003-9985.
- ↑ Sucheta Srivastava, M.D.. Breast - Noninvasive lobular neoplasia - LCIS classic (Differential diagnosis section). Topic Completed: 1 September 2017. Minor changes: 17 May 2021
- ↑ . Protocol for the Examination of Resection Specimens from Patients with Ductal Carcinoma In Situ (DCIS) of the Breast, Version: 4.4.0.0. Protocol Posting Date: June 2021. College of American Pathologists.
- ↑ Image annotation by Mikael Häggström, MD, using source image from:
Moatasim A, Mamoon N (2022). "Primary Breast Mucinous Cystadenocarcinoma and Review of Literature. ". Cureus 14 (3): e23098. doi:. PMID 35464581. PMC: 8997314. Archived from the original. .
- "This is an open access article distributed under the terms of the Creative Commons Attribution License CC BY 4.0."
Source for microinvasion: . Protocol for the Examination of Resection Specimens from Patients with Ductal Carcinoma In Situ (DCIS) of the Breast, Version: 4.4.0.0. Protocol Posting Date: June 2021. College of American Pathologists. - ↑ Tozbikian, Gary; Brogi, Edi; Vallejo, Christina E.; Giri, Dilip; Murray, Melissa; Catalano, Jeffrey; Olcese, Cristina; Van Zee, Kimberly J.; et al. (2016). "Atypical Ductal Hyperplasia Bordering on Ductal Carcinoma In Situ ". International Journal of Surgical Pathology 25 (2): 100–107. doi:. ISSN 1066-8969.
- ↑ Gary Tozbikian, M.D.. Breast - Ductal carcinoma in situ - DCIS. pathologyOutlines. Topic Completed: 20 May 2020. Minor changes: 6 May 2021
- ↑ . Ductal Carcinoma in Situ of the Breast. Stanford Medical School (2020-08-27).
- ↑ Image by Mikael Häggström, MD.
- Reference for most common definition of comedo necrosis by size:
- Harrison BT, Hwang ES, Partridge AH, Thompson AM, Schnitt SJ (2019). "Variability in diagnostic threshold for comedo necrosis among breast pathologists: implications for patient eligibility for active surveillance trials of ductal carcinoma in situ. ". Mod Pathol 32 (9): 1257-1262. doi:. PMID 30980039. Archived from the original. . - ↑ Image by Mikael Häggström, MD. References for features:
- . Ductal Carcinoma in Situ of the Breast. Stanford Medical School (2020-08-27).
- Hayward MK, Louise Jones J, Hall A, King L, Ironside AJ, Nelson AC (2020). "Derivation of a nuclear heterogeneity image index to grade DCIS. ". Comput Struct Biotechnol J 18: 4063-4070. doi:. PMID 33363702. PMC: 7744935. Archived from the original. . - ↑ Hayward MK, Louise Jones J, Hall A, King L, Ironside AJ, Nelson AC (2020). "Derivation of a nuclear heterogeneity image index to grade DCIS. ". Comput Struct Biotechnol J 18: 4063-4070. doi:. PMID 33363702. PMC: 7744935. Archived from the original. .
- ↑ 11.0 11.1 Allred, D. C. (2010). "Ductal Carcinoma In Situ: Terminology, Classification, and Natural History ". JNCI Monographs 2010 (41): 134–138. doi:. ISSN 1052-6773.
- ↑ Laura C Collins, MD, Christine Laronga, MD, FACS, Julia S Wong, MD. Ductal carcinoma in situ: Treatment and prognosis. UpToDate. Literature review current through: Sep 2022. | This topic last updated: Aug 19, 2022.
- ↑ Top and bottom left images by Mikael Häggström, MD. Bottom right image from:
Kulka J, Madaras L, Floris G, Lax SF (2022). "Papillary lesions of the breast. ". Virchows Arch 480 (1): 65-84. doi:. PMID 34734332. PMC: 8983543. Archived from the original. .
- "This article is licensed under a Creative Commons Attribution 4.0 International License"
Image sources
- ↑ 1.0 1.1 Image(s) by: Mikael Häggström, M.D. Public Domain
- Author info
- Reusing images